Drug name: linvencorvir
CAS: 1808248-05-6
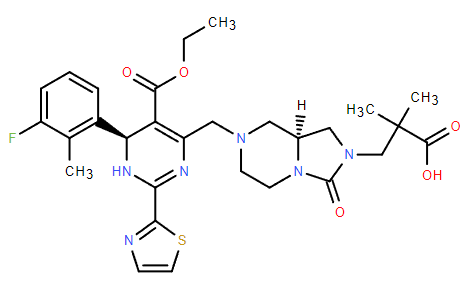
R&D code: RG7907,RO7049389
CAS: 1808248-05-6
Indications: Hepatitis B
Company: Roche
Phase 2
Results of a multicenter, non-randomized, uncontrolled open-label phase 2 clinical trial.
The study consisted of three cohorts: Cohort A) virally suppressed patients with chronic hepatitis B treated with Linvencorvir (600 mg orally once daily on an empty stomach; hereafter, same as below) + Nucleoside (Acid) Analog (NUC) for 48 weeks; Cohort B) untreated patients with chronic hepatitis B treated with Linvencorvir for 4 weeks followed by Linvencorvir + NUC for 44 weeks; and Cohort C) untreated patients treated with Linvencorvir + NUC + Peg-IFN-α for 48 weeks. 44 weeks; Cohort C) untreated patients treated with Linvencorvir + NUC + Peg-IFN-α for 48 weeks.
Primary endpoint: proportion of patients with HBV DNA below LLOQ (20 IU/mL) and HBsAg-negative (<0.05 IU/mL) at 24 weeks after discontinuation. Secondary endpoints: HBV DNA and RNA below LLOQ, HBsAg and HBeAg negativity and serologic conversion of anti-HBs and anti-HBe, change from baseline in viral markers such as HBV DNA and RNA and viral antigen, adverse events and pharmacokinetics.
Results of the study
A total of 72 chronically hepatitis B virus-infected patients were enrolled as subjects in the study, with 32 virologically suppressed patients enrolled in Cohort A, 10 untreated patients enrolled in Cohort B, and 30 untreated patients enrolled in Cohort C. The total number of subjects enrolled in the study was 72. Sixty-eight of these completed the 72-week treatment study (4 discontinued study drug due to non-safety concerns).
No patients in this study met the functional cure endpoint of HBV DNA below LLOQ (20 IU/mL) and HBsAg negative (<0.05 IU/mL) at 24 weeks of drug discontinuation.
Adverse Events
AEs occurred in 69% of NUC-suppressed patients in Cohort A (22/32), 90% of untreated patients in Cohort B (9/10), and in all 30 untreated patients in C. Headache, fever, and elevated ALT levels were the most common AEs. Elevated ALT levels occurred predominantly in weeks 2-8 and resolved within 14 weeks and were not accompanied by bilirubin/indirect bilirubin elevations, except for one patient with NUC suppression who had mild bilirubin elevations. NUC-suppressed patient who developed mild bilirubin elevations who had prior liver disease (cholestasis and Gilbert's syndrome).
In addition, in all five patients who developed grade 4 ALT elevations, linvencorvir was discontinued according to the protocol, but no further ALT elevations were observed after re-dosing. The majority of AEs were grade 1-2. grade 3-4 AEs occurred in 4 patients in cohort A (13%), 2 patients in cohort B (20%), and 11 patients in cohort C (37%). there were 8 cases of severe AEs and 1 death (due to malignant melanoma) that were not related to linvencorvir. The majority of treatment-related AEs occurred in Cohort C: 74 were associated with linvencorvir, 25 with NUC, and 266 with Peg-IFN-α. In each of Cohorts A and B, 4 AEs were assessed as linvencorvir-related.
