Voxelotor, a drug for sickle cell disease
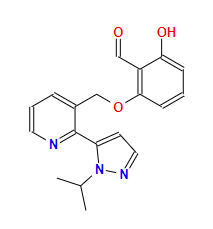
Name: Voxelotor
CAS: 1446321-46-5
Originating company: Global Blood Therapeutics
Indication: Sickle cell disease
Time on Market
The FDA authorized Oxbryta (voxelotor) for the treatment of sickle cell disease (SCD) in adults and children patients 12 years of age and older on November 25, 2019.The European Commission (EC) approved the sale of Oxbryta (voxelotor) on February 16, 2022, for the treatment of sickle cell disease (SCD) in adults and pediatric patients 12 years of age and older, as well as for the treatment of hemolytic anemia brought on by SCD, either as a monotherapy or in combination with hydroxyurea. China, Japan, etc. don't yet have it available.
Sickle Cell Disease
A collection of genetic red blood cell diseases is known as sickle cell disease (SCD). Hemoglobin is an oxygen-carrying protein found in red blood cells. Round and healthy red blood cells use tiny blood capillaries to transport oxygen to different parts of the body. Red blood cells in persons with SCD have defective hemoglobin, which makes them hard and sticky and resemble the "sickle"-shaped farm implement. Red blood cells are constantly in insufficient supply due to the premature death of sickle cells. Additionally, they become caught and obstruct blood flow when they cross tiny blood veins. This may result in discomfort and other severe side effects, including infections, acute chest syndrome, and strokes.
Millions of people around the world suffer from sickle cell disease (SCD), which is more prevalent in people whose ancestors originated in sub-Saharan Africa, Saudi Arabia, India, and Mediterranean nations like Turkey, Greece, and Italy. It is also prevalent in people whose ancestors came from the Spanish-speaking countries of the Western Hemisphere (South America, the Caribbean, and Central America). Chem-medications are synthetic medications that are sold in public.
In the US, 1 in 365 Black or African-American babies are born with SCD, 1 in 16,300 Hispanic babies are born with SCD, and 1 in 13 Black or African-American babies are born with sickle cell trait (SCT). Synthetic drugs (Chem-Drugs) for public health.
API Supplier (USDMF)
Registration date: 3/30/2022
Company: CURIA SPAIN SAU
Drug ingredient: VOXELOTOR
Synthesis route
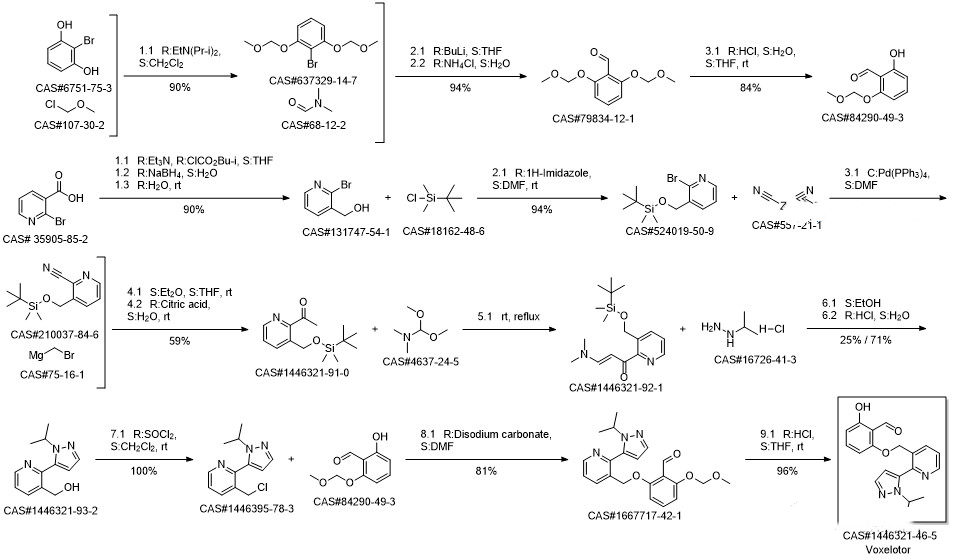

Reference 合成药物


