Chemical Name: Zasocitinib
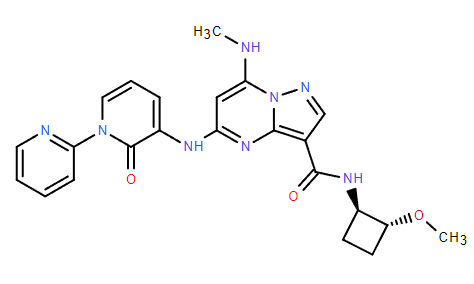
CAS: 2272904-53-5
R&D Code: TAK-279, NDI-034858
Company: Takeda
Phase III: Plaque psoriasis
On March 18, 2023, Takeda announced positive results from a Phase 2b study (NDI-034858) of investigational TAK-279, a drug that has been used in the treatment of psoriasis.
In the study of Phase 2b, 259 patients were randomly assigned (1:1:1:1:1:1 ratio) to receive one of four doses of TAK-279 once daily or placebo for 12 weeks. Results showed.

A significantly higher proportion of TAK-279 patients achieved a psoriasis size and severity index of 75 (44%, 68%, 67%; 5mg, 15mg, 30mg, respectively) versus placebo (6%; p < 0.001), consistent with the primary endpoint of the study.
A significantly higher proportion of TAK-279 patients achieved a psoriasis size and severity index of 90 (21%, 45%, 46%; 5mg, 15mg, 30mg, respectively) versus placebo (0%; p<0.001), and a psoriasis size and severity index of 100 (10%, 15%, 33%; 5mg, 15mg, 30mg, respectively) versus placebo ( 0%; p < 0.001 at 30 mg).
After 12 weeks, the TAK-279 group had a significantly higher proportion of patients with an overall physician assessment score of 0 ~ 1 than the control group (27%, 49%, 52%; 5 mg, 15 mg, 30 mg, respectively) or 0 (10%, 15%, 33%; 5 mg, 15 mg, 30 mg, respectively) and placebo (4% [p ≤ 0.001] and 0% [p<0.001], respectively). A weekly physician assessment score of 1 meant that the skin was essentially clean, while a score of 0 meant the skin was entirely clean.
Psoriasis area and severity index and overall physician assessment response rates (18%, 2%, 10%, 2%; psoriasis area and severity index 75, psoriasis area and severity index 100, overall physician assessment 0/1, overall physician assessment 0) in the TAK-279 2mg arm did not differ statistically significantly from the placebo.

